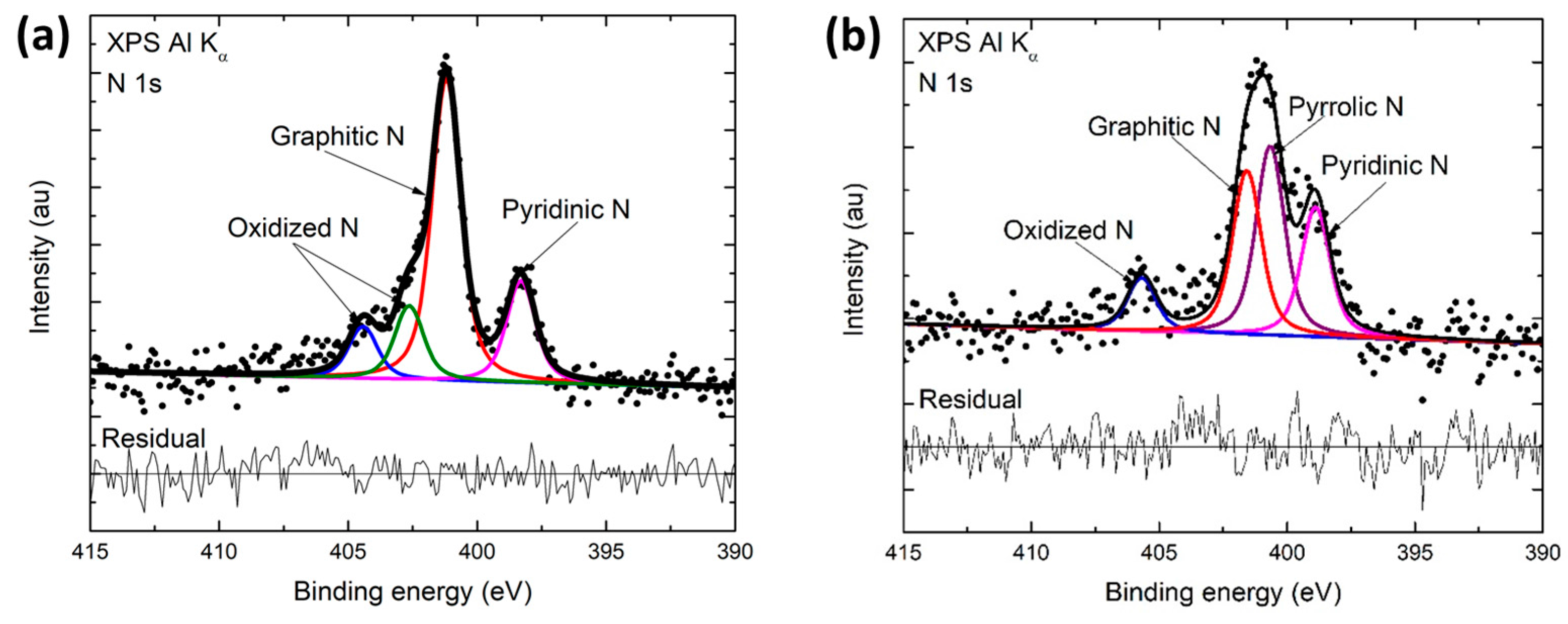

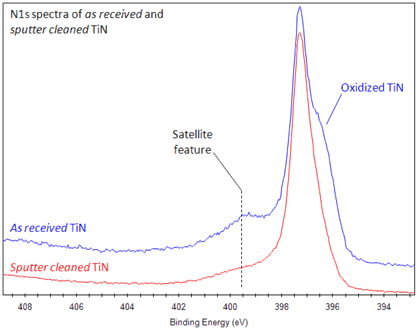
Some instruments have peak identification features, but otherwise, the identification of peaks/lines on the spectra can be completed by looking at standards of different materials. The term refers to various techniques, depending on whether the ionization energy. Electrons in 2s will have greater energy than those in 2p. Photoemission spectroscopy (PES), also known as photoelectron spectroscopy,1 refers to energy measurement of electrons emitted from solids, gases or liquids by the photoelectric effect, in order to determine the binding energies of electrons in the substance. peaks from electrons in 1s will have a greater energy than peaks representing electrons from 2s. The greater the binding energy, the greater the attraction of that electron to the nucleus. These changes in the nitrogen environment can produce significant differences in the nitrogen 1s XPS spectra region. As seen in the figure above, the O1s has the largest peak, and it shows that the atomic composition of oxygen is the greatest. Therefore, the peak intensities give information about the percent composition of a material. For example, if a peak, A, is half the height of another peak B, that means there were half as many electrons detected with the binding energy at A compared to the number of electrons detected with the binding energy at B. The shorter the peak, the less electrons represented. Peaks from the XPS spectra give relative number of electrons with a specific binding energy. References 1 AV Shchukarev and DV Korolkov, CEJC 2(2. Remainder of intensity under at Ru3d 3/2 energy can then be attributed to carbon. Evolution of Al2p peak, as measured by XPS, as a func-tion of nitrogen pressure (a) 0 mTorr, (b) 5 mTorr, (c) 10 mTorr, (d) 15 mTorr and (e) 30 mTorr. Intensity and binding energy of Ru3dp 5/2 peak is set by the Ru3d 3/2 peak, according to the spin-orbit splitting and ratio. (Figure courtesy of the creative commons license. Use Ru3d 5/2, which does not overlap with C1s, as a guide for any peak fitting.
Graph showing the binding energies of electrons from different orbitals (F1s, O1s, Si2p, etc.) and their intensities which tell the atomic composition of the sample based on the amounts of each electron from different orbitals present. Electrons of different energies follow different paths through the detector which allows the computer to differentiate the electrons and produce the spectra seen below.įigure \(\PageIndex\): XPS spectra. The energy of those ejected electrons is analyzed by a detector and a plot of these energies and relative numbers of electrons is produced. Based on these results and the results reported in the reviewed literature a picture of the microstructure of carbon nitride deposited using energetic species is developed.X-rays (photons) are shot onto a sample, and when electrons in the sample absorb enough energy, they are ejected from the sample with a certain kinetic energy. Furthermore, both deposition at elevated temperatures (350 ☌) and post-deposition ion irradiation have a strong influence on the bonding configuration in the CN films. For samples deposited at elevated temperatures an ordering of the amorphous CN network towards a crystalline graphitelike structure is observed. The core-level spectra of samples deposited at room temperature show that nitrogen is incorporated into the amorphous network in two different bonding configurations carbon has three main bonding configurations whose relative contributions vary as a function of the nitrogen content. This paper reviews x-ray photoelectron spectroscopy studies on carbon nitride (CN) and reports on results obtained from CN thin films prepared by mass selected ion-beam deposition.
